
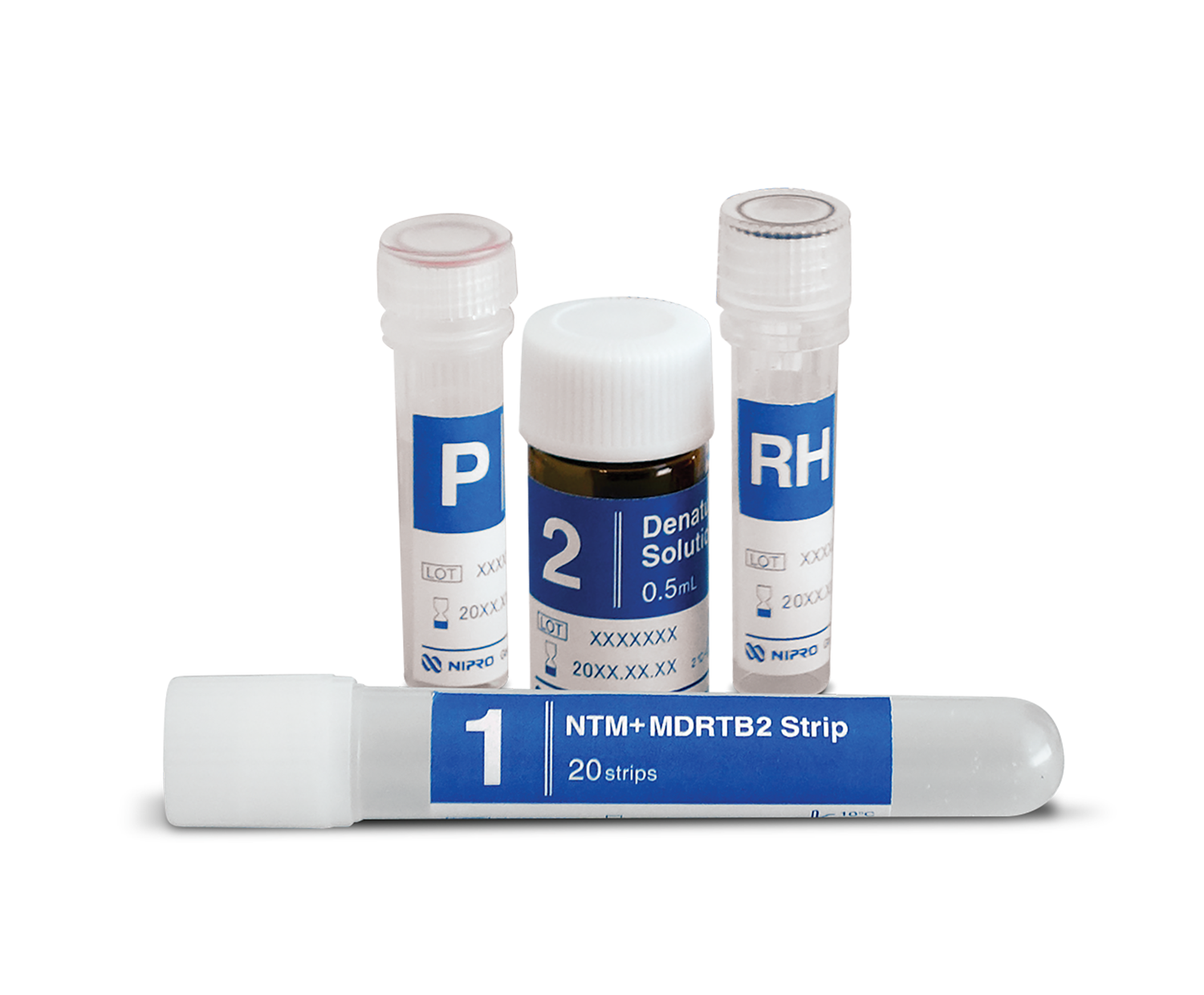
Drug susceptibility testing for tuberculosis based on line probe assay.
Genoscholar™ NTM+MDRTB II is approved by the World Health Organization (WHO) and distinguishes between Mycobacterium species while also testing resistance to rifampicin and isoniazid.
- Identification of Mycobacterium species
- Detection of resistance to rifampicin and isoniazid, based on gene mutations
- Sample preparation from sputum or culture
- Test results within 1 day
Specifications
- Kit contains necessary solutions/reagents for DNA amplification and DNA detection steps
- Manual analysis or automated analyzer (Multi-Blot NS-4800)
For more information, we invite you to contact our IVD team.
Need help?
Get in touch
Have questions or need assistance with our products? Feel free to reach out to our dedicated team.
| Give us a call +32 15 263 500 |
E-mail us [email protected] |
|
Submit a detailed inquiry: |
View our manufacturing locations: |


