Optimized filling-line performance and drug-container compatibility!
ENHANCE vials manufactured with the Nipro proprietary VIALEX™ thermal surface treatment combine premium quality with an exceptional inner surface durability to meet the requirements of complex medicines for injection.
-
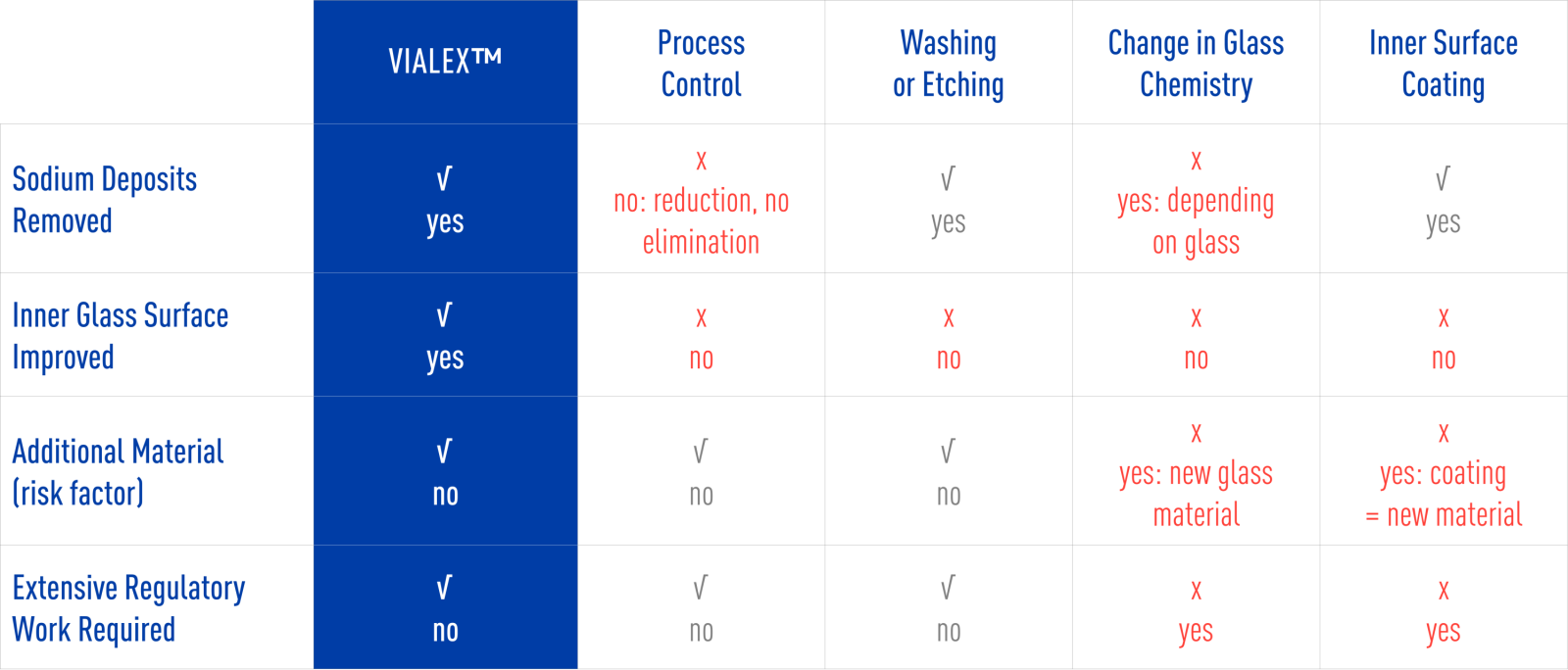
No extra materials nor change in glass chemistry.
-
Mitigates the effects from the converting process twofold: Sodium concentration reduced and inner glass surface improved.
-
VIALEX treated vials offer low levels of extractables & leachables, reduced alkalinity at the glass surface, and enhanced chemical durability.
-
Test results show less interactions of drug molecules and formulations with the inner glass surface.

Optimized Filling-line Performance
-
ENHANCE glass vials are highly consistent in size and shape. Each vial undergoes meticulous 100% camera inspection and in-line security for a wide range of dimensional parameters. The precise dimensions support reliable handling during the fill-finish process.
-
Crafted from top-of-the-line Type I, borosilicate glass tubing, ENHANCE vials offer an extremely consistent wall thickness. This quality goes hand in hand with reduced glass-to-glass contact throughout the entire vial production process, resulting in increased mechanical durability.
-
The in-depth, 100% in-line camera inspection extends to multiple cosmetic parameters. As a result, ENHANCE vials demonstrate an excellent cosmetic quality that minimize the risk of false rejects at final inspection.
Meeting the Highest Regulatory and Quality Requirements
- Very stringent AQLs regarding hydrolytic resistance, dimensional and cosmetic attributes, contamination, and glass defects reflect the exceptional container quality of ENHANCE vials.
- Production takes place in controlled environments and the automated packing process is in ISO-class cleanrooms, both supporting low levels of particles.
- ENHANCE vials are designed to meet the requirements of global Pharmacopoeias, including those of Europe, the United States, and Japan, and conform to ISO 8362-1.

The inner glass surface of the vial undergoes Nipro's proprietary thermal surface treatment, known as VIALEX™, which involves no additional materials or alteration in glass chemistry and is subject to 100% in-line inspection. This treatment mitigates the effects of the converting process in two ways: by reducing sodium concentrations and by improving the inner glass surface.
For Biotech
- Less interactions of drug molecules and formulations with the inner glass surface
- Optimized lyophilization process (less fogging)
- Reduced risk of glass delamination
For Diluents
[Water for injection | Aqueous NaCl solution]
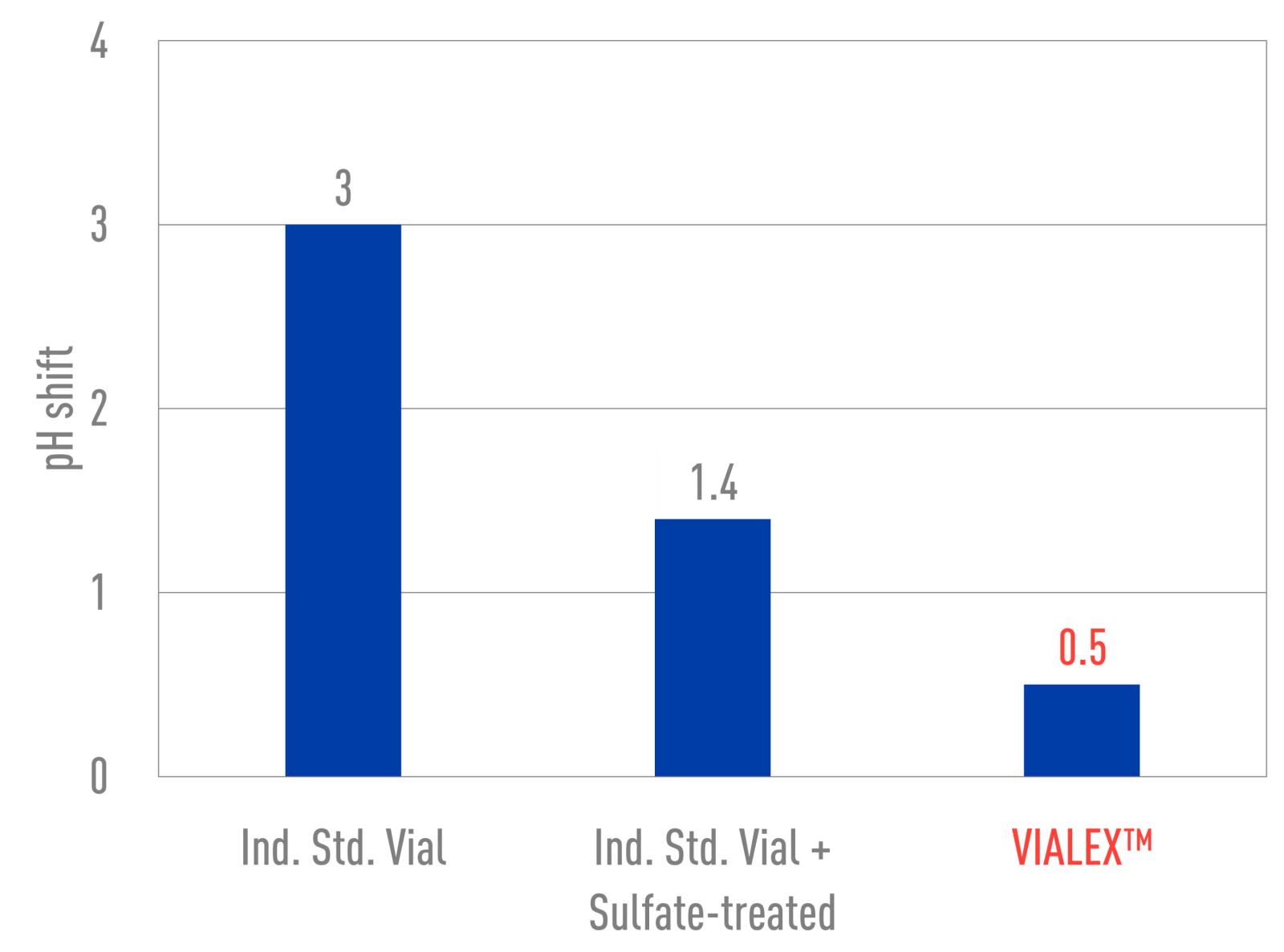
- Lower pH shift
- Reduced risk of glass delamination


Low Levels of Alkalinity
USP <660> / EP 3.2.1: SURFACE HYDROLYTIC RESISTANCE
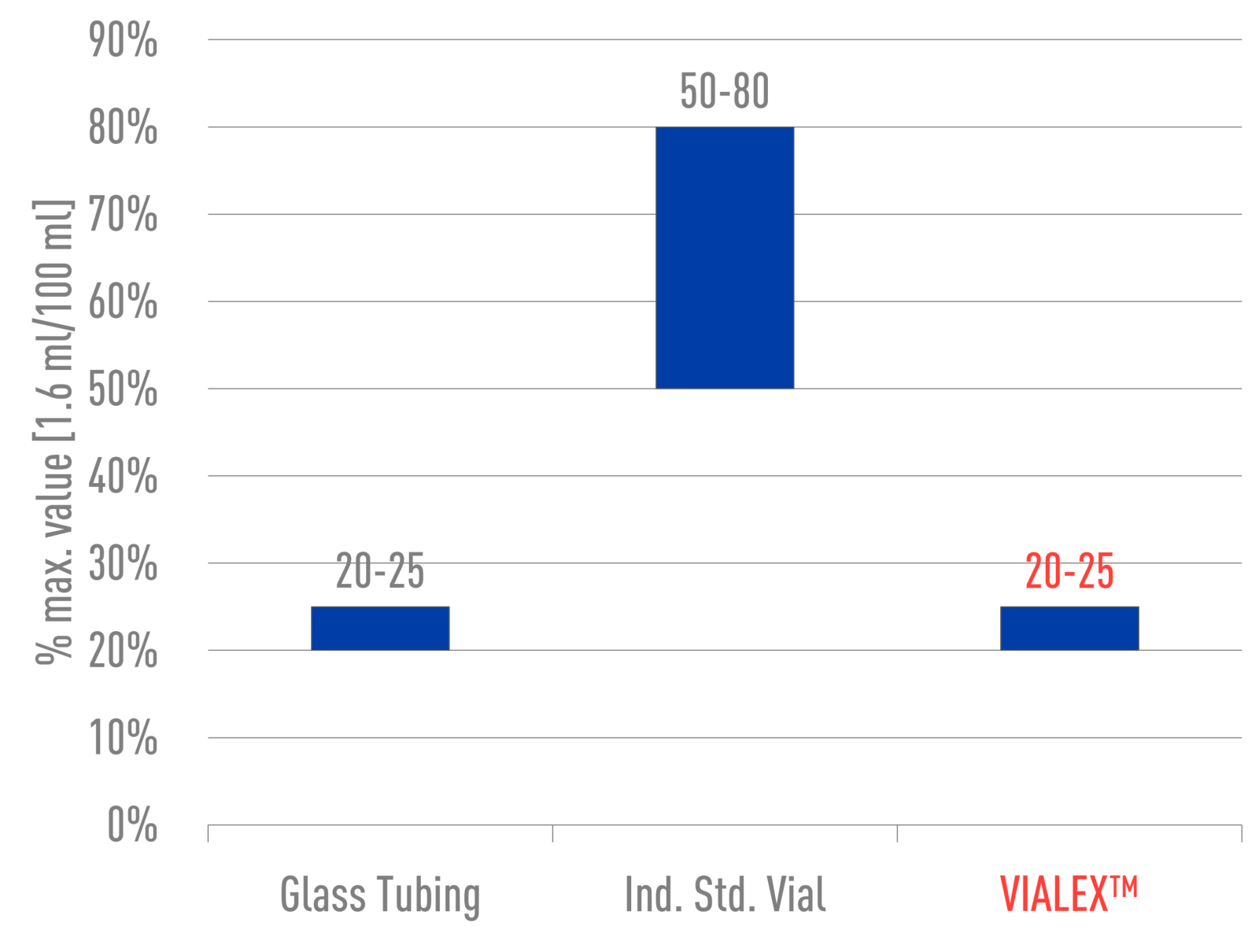
VIALEX-treated vials demonstrate an unprecedented surface hydrolytic resistance, comparable to that of original glass tubing. They consistently perform at ≤25% of the maximum limit.
2 ml, Type I, Class A, 51 Exp.
Enhanced Chemical Durability
LONG-TERM STABILITY STUDY | SEM: 24 weeks at 40°C – 51exp
For Phosphate, High Purity Water, Citrate, NaCl with Terminal Sterilization
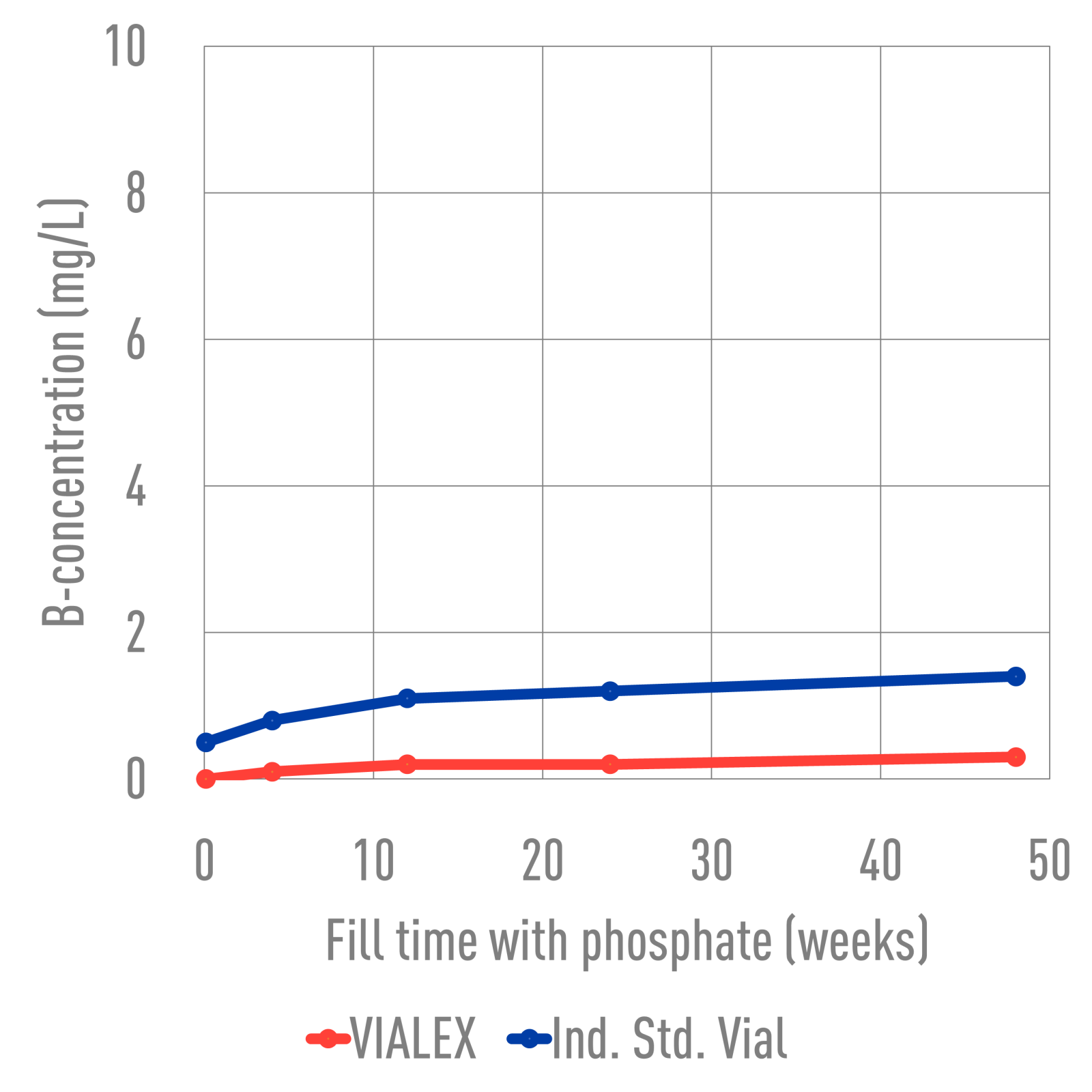
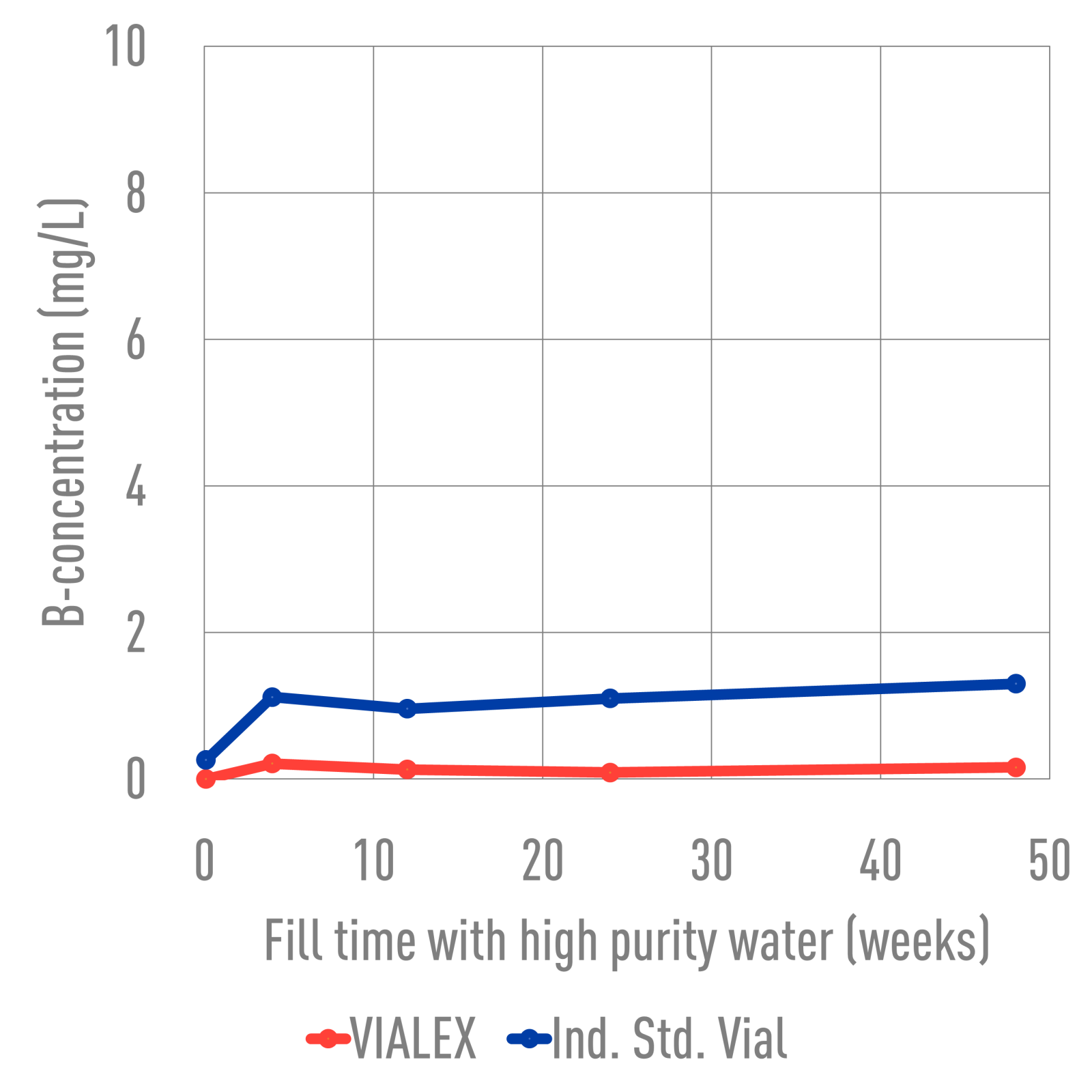
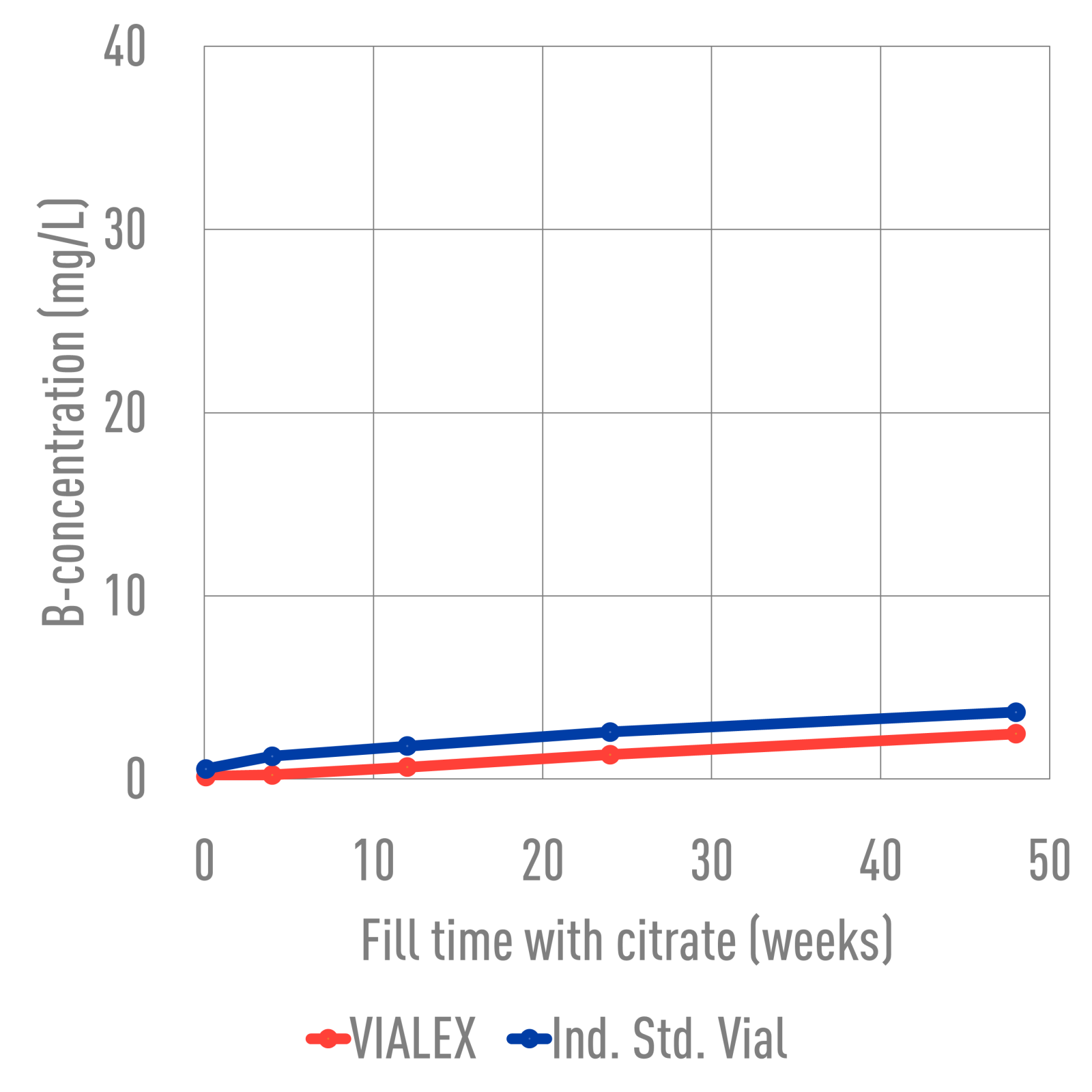
Ind. Std. vials and VIALEX-treated vials were filled with various buffer solutions and stored for 24 weeks at 40°C. SEM images were taken from the inner glass surface, specifically the heel area, to assess the degree of interaction between the vial inner surface and the buffer solution. The images were analyzed to determine if there was a difference in the degree of interaction between the two types of vials. VIALEX-treated vials show improved surface durability and fewer interactions compared to an industry standard vial.
Ind. Std. Vial

VIALEX™

Ind. Std. Vial

VIALEX™

Ind. Std. Vial

VIALEX™

Ind. Std. Vial

VIALEX™

Low Levels of Extractables
LONG-TERM STABILITY STUDY | 24 weeks at 40°C – 51exp
For Phosphate, High Purity Water, Citrate, NaCl with Terminal Sterilization
In terms of extractables, VIALEX-treated vials demonstrate lower levels compared to Ind. Std. Vials.
Silicone
Boron
Silicone
Boron
Silicone
Boron
Silicone
Boron
Minimal pH shift
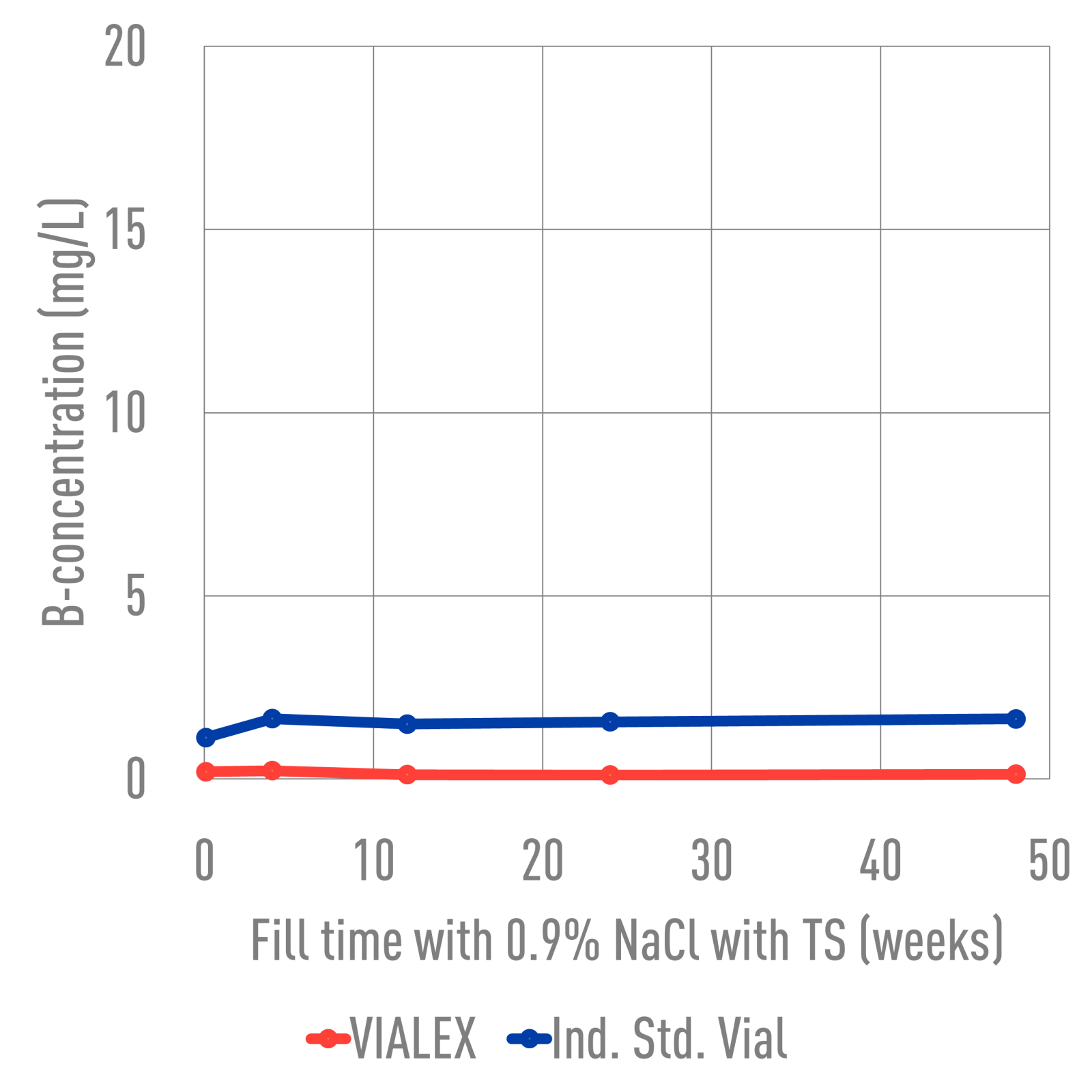
43 hrs. | at 121°C
NaCl 0.9% with Terminal Sterilization, pH at start 5.2
VIALEX-treated vials exhibit the lowest level of pH shift when compared to Ind. Std. vials and Ind. Std. vials + Sulfate-treated. While ammonium sulfate-treated vials appear to show an improvement in durability, the surface still has not been restored.
2 ml, Type I, 51 Exp.
| Glass Composition | Glass Type | Glass Expansion | Vial Type | Vial Sizes |
|---|---|---|---|---|
| Borosilicate | Type I | 51 | Crimp Neck | 2R |
| 33 | 4R | |||
| 6R | ||||
| 8R | ||||
| 10R | ||||
| 15R | ||||
| 20R |
Contact us for ENHANCE vials with VIALEX,
to improve your filling-line performance and drug-container compatibility!
For information regarding Nipro products, services, and resource material:
| Give us a call +32 15 263 500 |
E-mail us pharmapackaging@nipro-group.com |
| Submit a detailed inquiry: Contact per expertise |
View our manufacturing locations: Our global network |