For Drug Products Highly Sensitive to Silicone
Silicone oil can interact with highly sensitive molecules found in biotech products.1,2,3 Consequently, it has been recognized as one of the potential causes for aggregation and particle formation in therapeutic protein formulations.
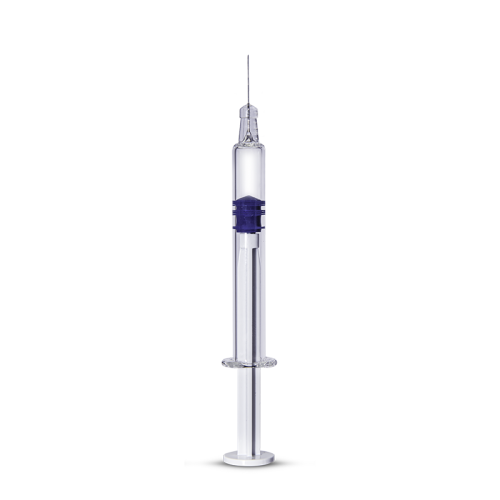
Nipro's D2F™ Pre-fillable Glass Syringe - Silicone Oil-Free is a solution for drug products highly sensitive to silicone oil. The key quality attributes that are impacted by the absence of silicone have been tested4 thoroughly and the results confirm the excellent performance of the D2F™ Pre-fillable Glass Syringes - Silicone Oil-Free:
-
Smooth Plunger Movement Performance
Consistent break loose and gliding forces due to plunger characteristics and hydrophilic glass surface with high surface free energy, good wettability, and good adhesiveness
-
Very Low Particle Levels
Amount of visible and subvisible particles significantly below the limit criteria stated in EP 2.9.20 and EP 2.9.195
-
Proven Container Closure Integrity
The syringes are offered in ENHANCE quality to meet the challenging drug product and service requirements of the pharmaceutical industry for sensitive drug products.
-
Excellent Drug-container Compatibility
-
Smooth Integration into Auto-injection Devices
-
Optimized Processability for Minimal Drug Loss
-
Extended Data Package for Fast Acceptance of Goods
| Volume (ml) | Outer Diameter (mm) | Tip | Flange | Shoulder |
|---|---|---|---|---|
| 1.0 long | 8.15 | Staked needle | Round | Round |
| Luer lock | Small round | |||
| Cut |

D2F™ Sterile Packaging
D2F™ syringes are manufactured in technologically advanced production sites that are certified according to ISO 15378, ISO 14001, and ISO 50001. Our fully automated cleaning and packaging process takes place in an ISO 7 / ISO 8 cleanroom with 100% monitoring under laminar air flow.
Special emphasis is placed throughout the entire D2F™ production process on particle reduction:
- Tub and nest are cleaned with ionized air
- No glass-to-glass contact
- Syringes are carefully washed
- Under laminar air flow
Nipro’s D2F™ packaging design is a smart and efficient choice enabling direct use on the filling line.
References
- Latoya S. Jones, Allyn Kaufmann, C. Russel Middaugh, Silicone Oil Induced Aggregation of Proteins, Journal of Pharmaceutical sciences, vol. 94, no. 4, April 2005.
- Michael Adler, Challenges in the development of pre-filled syringes for biologics from a formulation scientist´s point of view, American Pharmaceutical Review, 2012.
- Tina Tölke, Jenny Rudolf, Andreas Pfuch, Bernd Grünler, Syringe Siliconization, Pharm. Ind. 81, Nr. 3, 404-409, 2019.
- D2F Pre-fillable Glass syringes - Silicone Free - Nipro Product Development Report 2023.
- Compared to standard silicone oil syringe system
Frequently Asked Questions (FAQs)
"PFS" stands for "Pre-Filled Syringe." A pre-filled syringe is a type of medical device used for the storage and administration of medications. It consists of a syringe barrel pre-filled with a specific dosage of medication, a plunger, and needle or needle cap. Pre-filled syringes are designed to be used immediately without requiring additional preparation or assembly by healthcare professionals.
Pre-filled syringes offer various advantages when compared to other container types:
- Accurate dosing
- Convenience and ease of use
- Reduced risk of contamination
- Improved patient safety
- Minimized medication wastage
- No reconstitution is required
Yes, silicone oil-free syringes do exist. These syringes are manufactured using materials and processes that eliminate the presence of silicone oil, which can be a concern for certain pharmaceutical applications.
Direct 2 Fill (Direct to fill) Prefillable Syringes is the brand name of Nipro Pharmapackaging Ready to Use syringes.
Prefilled syringes offer several advantages, including reduced medication errors, minimized waste, improved dosing accuracy, and enhanced safety for both healthcare providers and patients. They eliminate the need for drawing medication from a vial, thereby reducing the risk of contamination and dosing errors. Additionally, they are convenient and allow for easier, more efficient administration, especially in emergency situations or for self-administration by patients with chronic conditions.
Prefilled syringes have significantly improved the ease of self-administration for patients, particularly those with chronic conditions that require regular injections, such as diabetes or rheumatoid arthritis. These syringes often come with features designed to make self-injection easier and safer, such as autoinjectors, safety needles, and ergonomic designs. However, it is recommended to perform functionality tests to prove the complete compatibility of PFS with autoinjectors, and patient education is crucial to ensure proper use and handling of these devices.
The environmental impact of prefilled syringes is a topic of ongoing discussion. PFS can reduce medication waste and the overall volume of materials used by eliminating the need for separate vials and syringes. On the other hand, the single-use nature of these devices raises concerns about plastic waste. Some manufacturers are addressing these issues by developing more sustainable materials and recycling programs.
The functionality and performance of our D2F PFS is maintained down to -40°C. Please contact our technical customer service for further advice on your drug development project.
PFS are answering drug-container interaction and administration challenges in the following therapeutic areas: Infectious diseases, Oncology, Cardiovascular Diseases, Chronic Diseases, Animal Health.
One of the primary reasons for opting for silicone oil-free syringes is to reduce the risk of protein aggregation in biologic drugs. Proteins are particularly sensitive to surface interactions, and the presence of silicone oil droplets can lead to protein aggregation, which can significantly alter the therapeutic effectiveness of a drug and may provoke unwanted immune responses in patients. Nipro has developed a D2F PFS silicone-oil free system.
For information regarding Nipro products, services, and resource material:
| Give us a call +32 15 263 500 |
E-mail us [email protected] |
| Submit a detailed inquiry: Contact per expertise |
View our manufacturing locations: Our global network |